-
-
Access Free Student Software
Ansys empowers the next generation of engineers
Students get free access to world-class simulation software.
-
Connect with Ansys Now!
Design your future
Connect with Ansys to explore how simulation can power your next breakthrough.
Countries & Regions
Free Trials
Products & Services
Learn
About
Back
Products & Services
Back
Learn
Ansys empowers the next generation of engineers
Students get free access to world-class simulation software.
Back
About
Design your future
Connect with Ansys to explore how simulation can power your next breakthrough.
Free Trials
ANSYS BLOG
December 16, 2020
Repairing Bone Loss with Simulation-Generated, Patient-Specific Implants
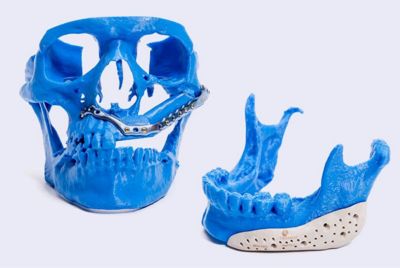
When a person loses bone structure due to an accident or a disease, it’s important for a surgeon to be able to reconstruct the lost bone as quickly as possible. Until recently, the surgeon might have a few titanium or plastic implants in different sizes to choose from, and they had to make them work, even if the sizing was not ideal. In the case of mechanical joints like the lower mandible (jaw), a good fit determines the functionality and quality of life of the patient.
Now Techfit Digital Surgery of Daytona Beach, Florida, has developed a patient-specific way of 3D-printing medical devices using computerized tomography (CT) and magnetic resonance imaging (MRI) scans of the patient to determine the perfect geometry for each implant. They use Ansys Discovery 3D design software to quickly develop the implant’s optimal design. Then they manufacture the implants, mostly using metal or plastic additive manufacturing, and deliver them for surgery within 10 working days.
“With Discovery, we know that our digital models will behave anatomically and mechanically like the patient will,” says Mauricio Toro, cofounder and CEO of Techfit. “It allows us to do very rapid iterations and improve the surgical product and surgical outcome.”
A 3 Step Process for 3D Printing Medical Devices
1. Acquiring Data
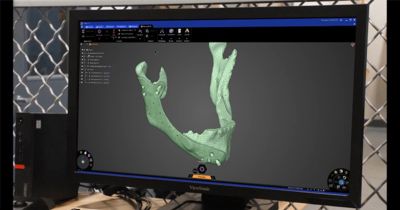
The first step in the surgical implantation process is to acquire data, which involves obtaining CT and MRI scans of the affected area. These scans are the starting geometry for the design. But as every engineer knows, geometries have to be cleaned up and modified before they can be useful.
Toro’s first big challenge was to find software that could input the STL file created from the scans and enable him to edit the geometry. He started using Ansys SpaceClaim first, with good results. But then along came Ansys Discovery.
Download a free trial of Ansys Discovery 3D design software.
“Ansys Discovery is a perfect match because it can process the STL files, which not every CAD package can handle, and mesh the geometry and make alterations in near real-time,” Toro says. “Discovery’s mechanical solver turns out a design within hours as opposed to days or weeks, which we don’t have because a patient is waiting for that implant.”
2. Co-creation and Validation
This second step in the process starts with the physician. Toro and his team meet with the surgeon to learn about his or her needs for a particular patient. They often take Discovery along with them for these meetings to show the surgeon their preliminary design based on the scans and make modifications from there.
“Especially if I have a case where I'm uncertain about the mechanical behavior of the bone or the implant, in a meeting with the surgeon I can start tweaking the model using Discovery,” Toro says. He explains that any implant design is always a compromise between the space you have and the implant’s mechanical properties. In many non-biological applications, you can just add more material to increase the factor of safety. But when you are implanting something in a human body, your packaging requirements are very strict.
“So, being able to perform simulations live with the surgeon and interact with them in a very fast feedback loop to understand where the sweet spot lies between mechanical performance and packaging is key to providing the best results achievable,” he says.
Toro finds this rapid feedback loop to be of critical importance.
“The saying goes that ‘practice makes perfect,’” he says. “But without a good feedback loop you can actually practice the wrong thing and become very good at doing something wrong. That’s why we’d rather say ’iteration makes perfection.’”
3. Manufacturing
Techfit currently manufactures all of its implants at company headquarters in Daytona Beach. They have all the standard machine tools needed for conventional manufacturing, along with additive manufacturing capabilities for more exotic implant shapes. Titanium alloys are most commonly used for metal implants. Plastic implants use PEEK for load-bearing applications and ultrahigh molecular weight polyethylene for tribological surfaces like joint replacements, where friction and wear can be a concern. Each implant is delivered with a mechanical validation analysis report so the surgeon has the utmost confidence in the surgical outcome.
The future of manufacturing at Techfit will include an additive manufacturing network distributed around the world, as Toro envisions it. A major reason for such a network is to deliver implants faster. If you make an implant in the United States for a patient in Saudi Arabia, that’s two days lost to shipping and another two days clearing customs, Toro explains. “If we have 10 days from CT scan to surgery, this means we are already four days late or our design time is crunched down into six days. Knowing a print is going to work on machine across the world by sending an STL file is going to be critical.”
Healing People Around the World
To date, Techfit has provided implants for several thousand patients.
“We have helped in some very incredible surgeries,” Toro says. These include hemi-mandiblectomies, where patients have lost half of their jaw because of tumors or accidents; a sternum replacement using a plastic implant fixed to the ribs with titanium plates; and, most commonly, thousands of cranial reconstructions required because of motorcycle accidents, tumors or head trauma.
Techfit also publishes papers in medical journals, presents results at conferences and even holds its own digital surgery conference annually.
“Custom surgery is actually a new field so it's not only a matter of making the implants but also a matter of evangelizing and ensuring adoption of the technologies,” Toro says. “Our goal is to make custom treatments the new standard for every patient around the world.”
Ansys simulation will continue to play a major role in achieving this goal.
“Having Ansys Discovery in our workflow allows us to validate our products faster, helps people get their surgeries faster and helps them recover faster,” Toro says. “One of the most rewarding things is you can actually see people's lives get better every time.”
Download a free trial of Ansys Discovery 3D design software.