Digitally Transforming Drug Manufacturing and Drug Delivery
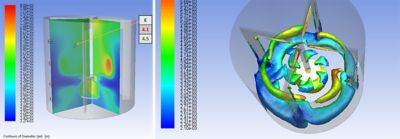
Traditional drug manufacturing and drug delivery process development require considerable time and financial investment. To accelerate innovation and dramatically reduce costs and time to market, in silico approaches are the only viable solution. Applying engineering simulation and mechanistic modeling to identify the best delivery route or to scale up production equipment from lab scale to mass production is crucial to achieve regulatory approval on a sustainable timescale and budget.