In Silico Technologies in Healthcare
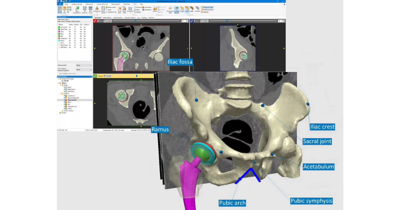
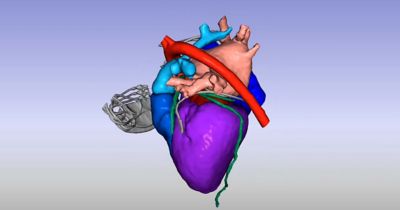
Medical solutions have advanced tremendously since the advent of modern medicine and continue to evolve with the help of in silico methods such as computational modeling and simulation (CM&S), artificial intelligence (AI), and digital twins. These technologies reshape the way healthcare professionals harness, process, and manage data, driving innovation and unlocking new possibilities for patients around the world.
Ansys, part of Synopsys, delivers the solutions, technology, and people to quickly scale up simulation deployment for many healthcare applications. With its multiphysics, multiscale, and multidisciplinary simulation platform and connected partner ecosystem, Ansys is uniquely positioned to increase efficiency and accelerate the innovation of next-generation healthcare solutions throughout the entire product life cycle.