-
-
Access Free Student Software
Ansys empowers the next generation of engineers
Students get free access to world-class simulation software.
-
Connect with Ansys Now!
Design your future
Connect with Ansys to explore how simulation can power your next breakthrough.
Countries & Regions
Free Trials
Products & Services
Learn
About
Back
Products & Services
Back
Learn
Ansys empowers the next generation of engineers
Students get free access to world-class simulation software.
Back
About
Design your future
Connect with Ansys to explore how simulation can power your next breakthrough.
Free Trials
Ever since the visionary father-son duo of Frederik and Gerard Philips started selling incandescent lightbulbs in the 1890s, their eponymous company has improved people’s lives with a steady flow of ground-breaking innovations.
Though consumers may know the company best for closer-shaving razors, deeper-cleaning toothbrushes, and the world’s first consumer video cassette recorder, Philips has been developing medical technology from the start. Among other achievements, its groundbreaking 1929 work on X-ray tubes made it possible to obtain quality images of organs, not just bones, enabling the early detection of tuberculosis and saving countless lives. Nearly a century later, the principle behind the original Philips “Rotalix” tube is still in use.
Today, Philips is focused exclusively on improving people’s health and well-being through meaningful innovations, and it aims to improve the lives of 2.5 billion people per year by 2030. As stated by its CEO, Roy Jakobs: “At Philips, we’re empowering people to take care of their health and well-being and helping doctors and nurses to provide better, more sustainable and more convenient care — in hospitals, clinics, and the home.”
The company is organized around three pillars:
- Diagnosis and treatment: supporting precision diagnosis and minimally invasive treatment in a growing number of therapeutic areas such as cardiology, peripheral vascular, neurology, surgery, and oncology.
- Connected care: connecting patients and caregivers across care settings, delivering clinical, operational, and therapeutic solutions.
- Personal health: broad range of consumer solutions to support people in proactively managing their health and well-being, including those ubiquitous Sonicare toothbrushes.
According to Ger Janssen, principal data and artificial intelligence (AI) scientist, Philips is leveraging what it calls “meaningful innovation” to optimize product performance and reliability, move away from expensive physical prototyping, and reduce time to market. That includes using Ansys multiphysics simulation software across a broad range of physics domains: heat and flow, mechanics, dynamics, optics, electromagnetics, acoustics, and electronics design. Philips deploys virtual design and testing for internal product development process acceleration and leverages the developed simulation models sometimes in the field of operational applications. The company is also working to integrate hybrid modeling — combining traditional simulation with AI — into its development capabilities.

A future hybrid modeling workflow using artificial intelligence to go from magnetic resonance imaging (MRI) to the cardiac simulation
First-time Success
Janssen says that to accelerate the product development process, Philips aims to virtually design as much as possible. That enables them to minimize hardware design cycles and design testing, and to eliminate problems before building physical prototypes.
“We’re looking for an appropriate trade-off between the virtual world and experimental input and validation, between detailed physical simulation and more simplified approaches,” says Janssen.
As an example, he points to the drop impact testing workflow developed by close cooperation of Janssen’s team with a few businesses, which proved its value across multiple Philips businesses.
In the traditional way, engineers simultaneously subjected physical prototypes to physical and virtual drop impact testing. The physical test would tell engineers if the prototype passed or failed, but the virtual test would tell them why. When a prototype failed to perform as planned, a new development cycle began.
Now, however, Philips has the ability to perform a simpler, shorter, and more effective strategy — a “first-time-right” approach that delivers detailed insight into impact dynamics and improves design confidence. Only after the product design “passes” the virtual drop impact testing, a prototype is made for physical verification. This means there are fewer hardware iterations, fewer tooling changes, and less testing, all of which generate considerable savings.
In a similar vein, Philips engineers use virtual qualification to understand how a product functions under different scenarios and stresses before they move to hardware qualification. This helps them make the right critical design choices early on, reducing cost, time, and uncertainty.

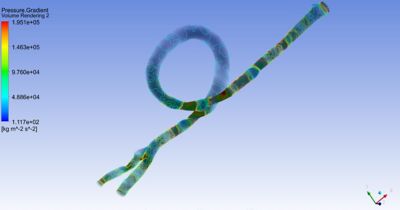
One way Philips uses simulation is in virtual design and qualification. The example above shows how clamping affects the mechanical stresses inside a catheter.
Digital Twins Model Patients
In addition to their role in research and development, Philips’ virtual design and qualification activities can also form the basis for digital twins in the operational and clinical space.
A digital twin is a virtual representation of real-world entities and processes, synchronized at a specified frequency and fidelity. Digital twins allow companies to monitor the real-time behavior of a system or product and compare it to a virtual model that is constantly being updated. This makes it possible to optimize performance, predict and avoid failure, and plan for maintenance of complex systems.
In the clinical space, a digital twin enables the dynamic combination of multiple models: a model of the patient’s anatomy plus computational and system models from the imaging device used to diagnose or treat their health issue. As a result, the clinician has unique insight into the patient’s current status, can test different scenarios, and can better predict the outcome.
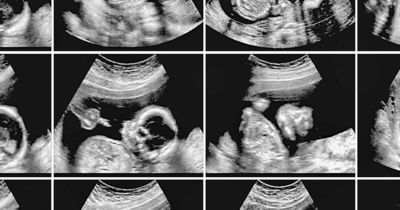
For example, to help clinicians diagnose structural heart problems, the Philips Dynamic HeartModel automatically extracts the anatomic detail of the patient’s heart from an ultrasound image and calculates again automatically some cardiac function parameters. The clinician is then also able to use the same technology in the planning and the performance of a cardiovascular intervention.
Ultimately, the digital twin — a virtual representation of a person’s health status — will enable clinicians to deliver the right care in the right way at the right time. It reduces patient adverse events, shortens hospital stays, and minimizes the likelihood of readmission.

Pre-operative risk assessment of paravalvular leakage using a computational transcatheter aortic valve replacement (TAVR) deployment model
Room for Limitless Innovation
Philips drives innovation and efficiency through a global, collaborative network where locally relevant solutions are created.
In the future, Janssen says, many of those solutions will come from the interaction of AI with traditional, knowledge-driven modeling and simulation. The process is already underway at Philips, in fact. It underscores Philips’ commitment to advancing highly personalized, digital healthcare through leading-edge technology — which is central to Philips’ strong healthcare legacy.
Learn more about how Ansys can help with your healthcare innovations.
Register for Simulation World to watch Philips' May 14 presentation "The Role of Modeling and Simulation," which is available on demand.
The Advantage Blog
The Ansys blog, featuring contributions from Ansys and outside experts, keeps you in the know with the latest industry information, including engineering articles and simulation news, thought leadership and trends, product development advances, tips to better use Ansys solutions, and customer stories.