ANSYS ADVANTAGE MAGAZINE
October 2021

Most people don’t appreciate their kidneys — until they fail. These small organs are responsible for filtering toxins from the blood and regulating electrolytes and blood pressure. They produce urine and critical hormones, and they help maintain the body’s pH balance. But in stage 5 kidney disease — also known as end stage renal disease (ESRD) — an individual’s kidneys have failed. They can no longer perform these critical functions adequately. Without intervention, death inevitably follows.
The options for intervention, though, are limited. A patient with ESRD can undergo dialysis, a clinical procedure in which a machine filters the blood and removes what toxins it can. Unfortunately, dialysis is only about 10% as effective as a kidney at detoxifying blood, and it provides none of the kidney’s metabolic benefits. Furthermore, dialysis comes at a price. The three- to four-hour procedure is expensive, and it can be so exhausting that patients tethered to a dialysis machine effectively lose the entire day. Nor is it just one day: Individuals on dialysis must undergo the procedure three times per week.
A kidney transplant offers a far greater benefit, as a transplanted kidney can perform all the functions of the patient’s original kidney. But too few kidneys are donated each year to meet the demand for transplants. At any given time, there may be more than 100,000 individuals on the kidney transplant waiting list, but only enough viable donated kidneys to perform around 20,000 transplants each year.
Creating the Kidney Project
All of these factors inspired The Kidney Project, a nationwide collaboration led by Shuvo Roy, Ph.D. of the University of California San Francisco and William Fissell, MD of Vanderbilt University Medical Center (VUMC), and involving collaborators at the University of Michigan and other institutions. The project’s goal has been to create a surgically implantable, self-sustaining bioartificial kidney that will perform as many of the functions of a healthy human kidney as possible.
One part of this tiny device — the hemocartridge — is designed to perform the blood filtration functions of the kidney. A second part — the biocartridge or bioreactor — is designed to house lab-grown cells from a human kidney that will deliver the metabolic benefits that a healthy human kidney would. In the first version of the bioartificial kidney, these embedded kidney cells (derived from human cadaver kidneys that were not viable for transplantation but whose tubule cells could be preserved, isolated and then replicated) are expected to support the regulation of electrolytes. In future versions, researchers working on The Kidney Project expect that other embedded kidney cells will add support for the regulation of pH and blood pressure as well as provide other hormonal benefits.
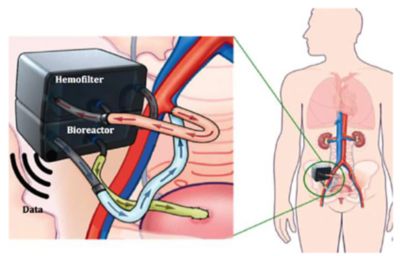
Representation of the implantable bioartificial kidney device showing the two-compartment structure. The patient’s blood passes through the hemofilter, where toxins are removed. The blood then redirects to the bioreactor where renal cells ensure proper reabsorption of water and salts from the ultrafiltrate back into the patient’s blood.
As designed, this tiny, surgically implanted device would operate continuously, powered not by batteries but entirely by the force of an individual’s blood pressure. Because toxin filtration would be ongoing, patients with a bioartificial kidney would not need to be connected to a dialysis machine three times a week. Nor would they experience the exhaustion that arises from dialysis. Such enhancements would have a powerful impact on an ESRD patient’s quality of life. Moreover, they would have a significant impact on Medicare spending. Medicare is US federal health insurance for certain people. ESRD is the only condition where the treatment is fully covered by Medicare regardless of the patient’s age. Today, Medicare spends more than $40 billion per year covering the cost of ESRD care. That’s around 7% of its annual budget, yet those funds care for only 1% of the Medicare population. No other single disease has this kind of disproportionate impact.

By refining designs and significantly reducing low shear regions, clot development was eliminated. The device on the bottom showed no clot formation after 30 days of implantation in an animal.
Building a Bioartificial Kidney
Using Ansys Mechanical, designers working with The Kidney Project created and refined detailed models of all the physical components of the bioartificial kidney, including the breakthrough silicon nanopore membrane technology that makes much of the bioartificial kidney operationally feasible. These membranes, created by The Kidney Project, encapsulate the lab-grown human kidney cells embedded in the device’s biocartridge. As blood flows across the membranes, the embedded kidney cells perform the same metabolic functions that the same cells would perform in a normal healthy kidney. After extensive stress simulations in Mechanical, the designers found the ideal balance of thickness, pore size and pore distribution for use in their design. The membrane’s micromachined pores are only 10 nm wide — too narrow for blood cells associated with the body’s immune system to penetrate the membrane and discover these “foreign” kidney cells, which could result in the body rejecting the bioartificial kidney. At the same time, the membrane’s pores are large enough to filter out the toxins, salts and water from the blood. Blood pressure continuously forces those filtrates through the hemocartridge and onto the biocartridge, which reabsorbs the necessary salt and water back into the blood, thereby concentrating the toxins into urine, which is directed onto the bladder.
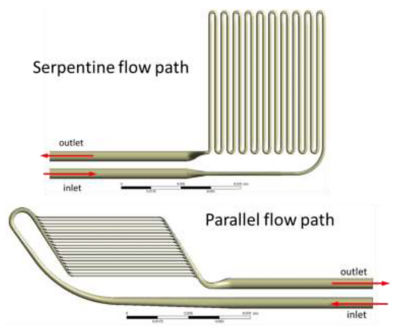
Candidate flow paths within the hemofilter compartment.
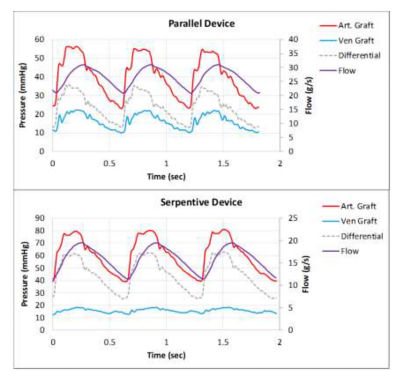
Measuring boundary conditions in vivo for candidate hemofilter flow paths with the computational fluid dynamics (CFD) output flow path indicated in purple
Given the need to flow blood across these membranes and through the channels within the device, researchers working with The Kidney Project knew that the device had to be designed to minimize the potential for blood clot formation. Clots can form when the chemistry of the blood reacts with a surface or when blood flow is disrupted and blood platelets congregate to form a nucleation site for a clot. Researchers knew that they could treat the surface of the silicon membrane with biologically friendly chemistries to prevent clotting caused by a chemical reaction, but needed a way to optimize and streamline blood flow though the device to avoid disruptions and ensure that blood flow remained laminar.
To accomplish this, engineers working with The Kidney Project teamed up with experts in computational fluid dynamics (CFD) at SimuTech Group, who used Ansys Fluent and Ansys CFX to simulate and analyze the flow of blood into, through and out of numerous bioartificial kidney designs. Researchers needed to understand where zones of blood recirculation might create clotting opportunities within a given design. They needed to tune the design of the filtration cartridge to keep the wall shear stress forces low enough to prevent red blood cells from exploding and becoming nucleation sites for clots. At the same time, they needed to ensure that design refinements did not reduce the low wall shear stress forces to a level so low (below 1 Pascal) that it could cause blood cells to congregate — in turn creating a nucleation site and conditions conducive to clot formation.
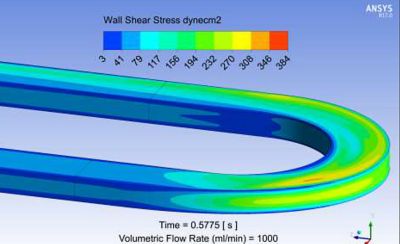
Full range wall shear stress contour plot from Ansys Fluent for a candidate bioreactor blood flow path.
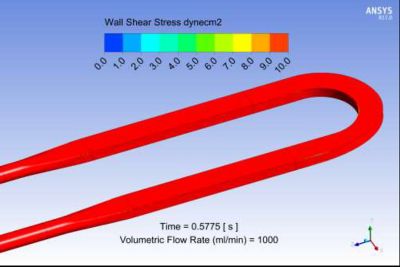
Limited range wall shear stress contour plot from Ansys Fluent for a candidate bioreactor blood flow path. The range is limited here to 10 dynes/cm2 ( or 1 Pa), which corresponds to the threshold clot nucleation value.
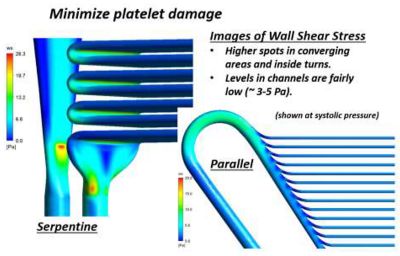
Ansys Fluent results of wall shear stress showing both low wall shear stress regions and high wall shear regions, which may lead to platelet damage or activation
Simulation Facilitates Design Filtration
Bringing the designs created in Mechanical into Fluent and then running blood flow simulations in Fluent made it easy for The Kidney Project team to assess and refine — to filter, as it were — a large number of design options. They could have created physical prototypes of each design from Mechanical, but testing each physical prototype would have been unfeasible. Such an approach would have taken far longer, incurred much higher costs and raised ethical issues because of the potentially high number of animals that would have been required to validate the effectiveness of the different prototypes.
By simulating the flow of blood through the different designs in Fluent and visualizing the outcomes in CFX, the researchers at The Kidney Project could measure blood flow velocity precisely, identify areas where clots were likely to form, understand why those spots created opportunities for clotting and then incorporate and test design adjustments. They could refine prototype designs quickly in Mechanical and then quickly run new simulations on the refined designs. This minimized the need to build and test prototypes on animals. Indeed, when the researchers at The Kidney Project did build and test a prototype of a promising design, they quickly discovered that blood clots formed in precisely the locations that Fluent had indicated they would — which prompted them to focus even more on refining the designs using Fluent prior to prototyping a promising candidate. They could see that there was strong correlation between the simulations and real-world outcomes.
Aiming for Incremental Improvements
While the goal of creating a bioartificial kidney may seem lofty, researchers at The Kidney Project understand that the device does not need to replicate the full functionality of a healthy human kidney perfectly. If it can provide patients with the level of kidney functionality that they had experienced at stage 4, it would change lives because many people with stage 4 kidney disease don’t even feel the downsides of kidney disease. They travel, they eat normally, they exercise; they live life in much the manner anyone else does. That all stops when dialysis begins and the long wait for a kidney transplant commences.
With help from Fluent and CFX, researchers at The Kidney Project are well on their way to delivering a bioartificial kidney that will fulfill its design goals — quickly, efficiently and far more cost-effectively than would otherwise be possible.
讓我們開始吧
如果您面臨工程挑戰,我們的團隊將隨時為您提供協助。憑藉豐富的經驗和對創新的承諾,我們邀請您與我們聯絡。讓我們共同合作,將您的工程障礙轉化為成長和成功的機會。立即與我們聯絡,開始對話。