ANSYS BLOG
July 6, 2021
Using Ansys for Doctorial Research in Cardiovascular Engineering
As a Marie-Curie fellow Benjamin Bhattacharya-Ghosh obtained his doctorate at University College London (UCL) under the supervision of Dr. Vanessa Díaz. Together with 12 other Marie-Curie fellow students, he has been a member of the European “Medical Devices and Design in Cardiovascular application” MeDDiCA project. Located in the UK, Italy, France, the Netherlands and Romania, all MeDDiCA students conducted their research in the field of cardiovascular engineering.
At UCL, Benjamin used Ansys CFD to couple a computational multiphysics and multi-scale model (MSM) of the left ventricle to a 3D model of a heart valve. The left ventricular model was used as a complex and multi-scale boundary condition (BC) for the 3D problem.
As one of four chambers in the heart, the left ventricle is of major importance, pumping oxygenated blood from the lungs to the entire body. The multi-scales involved in the MSM encompass the microscopic (protein and cellular) and the macroscopic (organ) scale, comprising typical characteristics and mechanisms at each scale, and representing the ventricular contraction and relaxation.
Benjamin Bhattacharya-Ghosh
Based on the cellular dynamics and protein kinetics, the MSM describes the ventricular haemodynamics at the organ scale, such as pressure, volume and flow of the blood entering and leaving the ventricle.
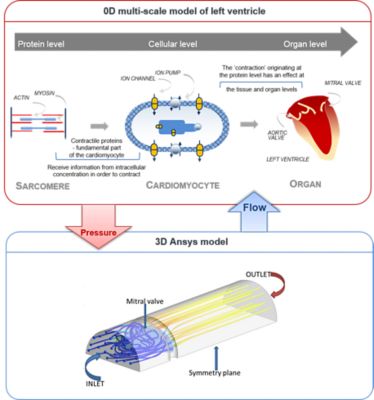
Figure 1: Coupling between 0D MSM and 3D Ansys model
The coupling to the 3D model of a heart valve was achieved through a collaboration with Dr. Justin Penrose and Dr. Santhosh Sheshadri from Ansys UK. The 3D model describes the distribution of bloodflow entering the left ventricle via the mitral valve (in this case represented by a mechanical heart valve), allowing a unidirectional bloodflow into the ventricle. Within this study, a novel technique has been developed that allows the continuous exchange of BCs between the 0D MSM and the 3D model. The BCs, presented in this 0D-3D coupling method, are defined by the ventricular pressure and the flow through the valve (Figure 1).
The pressure is calculated in the 0D model and subsequently provided to the 3D model, which applies the pressure value to calculate a corresponding flow value. Following, the flow value is supplied back to the 0D model to calculate the next pressure value.
The interaction and exchange of ventricular pressure and flow through the mitral valve between both models drives the local haemodynamics of the mitral valve in the 3D Ansys CFD model during the opening and closure of the valve, based on the physiological behaviour of cardiac cells within the 0D MSM.
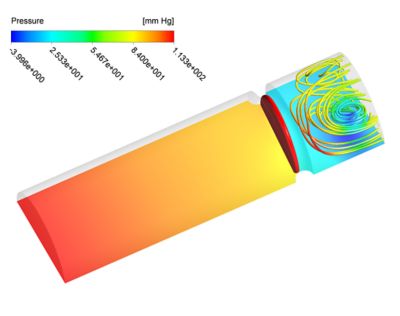
Figure 2: 0D-3D coupled simulation. Pressure contour plot along with pressure streamlines during contraction (left) and relaxation (right) of the ventricle and heart.
The simulations of the presented 0D-3D coupling have been performed using Ansys CFD, with the immersed solid method (ISM) option for the valve dynamics. The results (Figure 2) have shown a major improvement to prior simulations results of 0D-3D coupling techniques that have been presented in literature.
Previously reported simulation issues, such as mesh deformation and remeshing could be solved using the ISM option.
Ansys CFD has shown to be a useful tool to understand cardiovascular diseases.
The 0D-3D coupling sets a foundation and coupling technique to investigate other entities of the heart and conduct case studies on pathologies of the heart. Ansys software has been traditionally used in cardiovascular biomechanics but this novel implementation proves that unusual, diseased and malfunctioning conditions can be simulated (and treated) at the cellular level using the MSM, while the macroscopic implications and results can be reviewed from the effect on the valve function in the 3D Ansys model.
Cardiovascular diseases are still the leading cause of death worldwide. Simulations like this can help drug treatment evaluation in the future. This approach of using the MSM in combination with Ansys has shown to provide a method for gaining further insight into treatments at low computational costs.
Learn more about how simulation is being used for rapid and reliable innovation and testing of new cardiovascular medical devices and implants.