-
-
Software gratuito per studenti
Ansys potenzia la nuova generazione di ingegneri
Gli studenti hanno accesso gratuito a software di simulazione di livello mondiale.
-
Connettiti subito con Ansys!
Progetta il tuo futuro
Connettiti a Ansys per scoprire come la simulazione può potenziare la tua prossima innovazione.
Paesi e regioni
Customer Center
Supporto
Partner Community
Contatta l'ufficio vendite
Per Stati Uniti e Canada
Accedi
Prove Gratuite
Prodotti & Servizi
Scopri
Chi Siamo
Back
Prodotti & Servizi
Back
Scopri
Ansys potenzia la nuova generazione di ingegneri
Gli studenti hanno accesso gratuito a software di simulazione di livello mondiale.
Back
Chi Siamo
Progetta il tuo futuro
Connettiti a Ansys per scoprire come la simulazione può potenziare la tua prossima innovazione.
Customer Center
Supporto
Partner Community
Contatta l'ufficio vendite
Per Stati Uniti e Canada
Accedi
Prove Gratuite
A pioneering study at King’s College London is revolutionizing how clinicians predict and manage congenital heart disease (CHD) in unborn babies. Using advanced digital twin technology, researchers have developed an approach to improve the diagnosis of aortic coarctation (CoA) — a condition where the main body artery is narrowed, posing serious risks after birth.
Improving Diagnostic Accuracy for In-Utero Babies
CHDs are structural problems with the heart that are present at birth, affecting how blood flows through the heart and to the rest of the body, globally affecting over 4 million children under the age of 5 per year. Around two-thirds of all CHD-related deaths occur in children under 1. Prenatal diagnosis of CHD enables clinicians to more accurately plan the right treatment for babies as soon as they are born, improving both early survival and long-term outcomes for those affected. However, CoA, one of the most common types of CHD, is notoriously difficult to accurately predict, even with expert scans by fetal cardiologists.
King’s College London Research Department of Digital Twins for Healthcare is committed to developing digital health technologies to revolutionize how we understand and treat CHD and other complex diseases, such as blood disorders, diseases of the autonomic nervous system, and protein and insulin imbalances. The department combines multidisciplinary expertise from the fields of engineering, imaging, and medical sciences to build technology that improves patient care and outcomes on a global scale. Working in collaboration with the Research Department of Early Life Imaging, also based in King’s College, the team brings together clinicians, researchers, technologists, and communities in a dynamic environment where groundbreaking ideas can flourish and translate into real-world solutions.
The head of the Research Department of Digital Twins for Healthcare, Dr. Adelaide de Vecchi, focuses on the development of digital twins for precision medicine by combining imaging, machine learning (ML), and computational modeling and simulation (CM&S).
“Our vision is not only to enhance technological synergies but also to constructively engage all stakeholders, from engineers to clinicians, to policymakers, to patients, to enable the full potential of the digital twin paradigm and its deployment,” says Dr. de Vecchi.
Dr. de Vecchi specializes in precision cardiovascular research, specifically blood clot formation, pediatric CHD and aortic coarctation prediction, cardiac valve implantation, and ML-based assessment of pulmonary hypertension.
In 2022, Dr. de Vecchi and her colleague professor Pablo Lamata were given the opportunity to support Dr. David Lloyd and Dr. Kuberan Pushparajah from the fetal cardiology team at Evelina London Children’s Hospital to research CoA. Using simulation and imaging data, their team created a risk score assessment that doctors at Evelina Children’s Hospital currently use to help determine if a fetus is at risk for CoA.
Getting Ahead of the Problem
CoA is a CHD where a section of the aorta is abnormally narrowed, and it accounts for 7% to 8% of all CHD cases in newborns. Affected babies typically need surgery shortly after birth to fix the defect, making in utero detection essential for timely treatment. But accurately diagnosing the condition before delivery remains a significant challenge. Standard ultrasound methods often struggle to provide clear images due to fetal positioning and the complexity of the tiny vascular structure, resulting in false-positive rates of 50% to 90%.
These diagnostic inaccuracies not only burden healthcare systems but cause unnecessary stress for families as suspected CoA designates expectant mothers as high risk. High-risk pregnancies typically require additional monitoring, meaning more doctors' appointments, more costs, and more anxiety. After birth, newborns undergo extended hospital monitoring to confirm or rule out the condition.
This gap in accurate early diagnosis has led to growing interest in advanced imaging technologies and computational approaches to improve precision. Identifying CoA more reliably before birth could greatly enhance neonatal care by enabling clinicians to plan interventions and optimize outcomes.
“The idea was, can we actually make this diagnostic assessment more accurate and provide valuable insight to reduce false positives?” says Dr. de Vecchi.
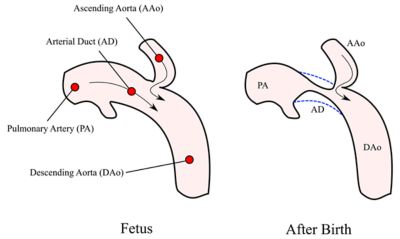
Coarctation of the aorta is a narrowing of the aorta after birth.
Simulating Fetal Cardiac Structures
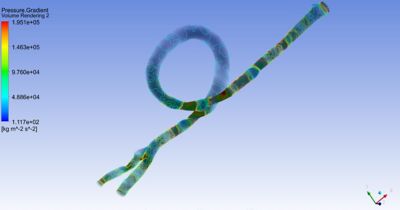
Using high-resolution, fetal cardiac magnetic resonance imaging (MRI), in-house shape modeling software, Ansys Fluent fluid simulation software, and Ansys Discovery 3D simulation software, the team created digital twins of the fetal aortic arch. These models enable the application of statistical shape modeling (SSM), a technique that analyzes the intricate geometry of the aortic arch in 3D. Unlike traditional two-dimensional ultrasound measurements, which often rely on subjective interpretation, SSM provides a standardized, data-driven analysis of vascular structures. This method enables researchers to identify subtle anatomical differences that may indicate CoA while accounting for natural variations in fetal development.
“We’re lucky that Evelina has a fetal MRI service, which is a step up from ultrasounds,” says Professor Lamata, “But it’s expensive to MRI every single woman, so our next step is to make 3D models based on the ultrasounds and then compare them to the MRI-based models we already have.”
While creating the SSM, the team noticed specific features that were present only in true CoA cases. Using anatomies from true CoA cases and false positives, the team created a virtual patient cohort and ran a large flow simulation study with Fluent software.
“We started to see some features in the flow that only happen in true CoA but not in false positives,” says Dr. de Vecchi. “Fluent really helped us understand the mechanism at play.”
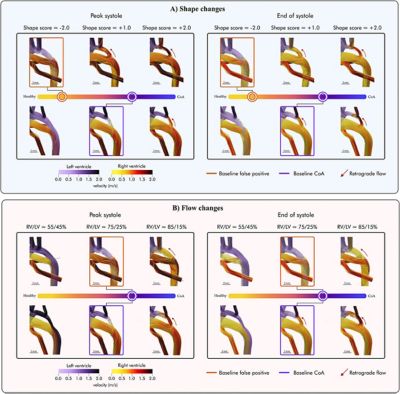
Panel A shows the impact of shape changes from the baseline simulations for the studied cases with representative phenotypes, false-positive (orange box) and true-positive coarctation of the aorta (purple box), spanning the disease spectrum captured by the shape score from healthy to diseased. From left to right, the anatomy changes from a healthier shape (yellow) to a more CoA one (purple). Panel B shows how variations in inlet boundary conditions influence the baseline hemodynamics (boxed) for the two studied shape phenotypes. The red arrows show the appearance of retrograde flow in the aortic isthmus (from Hermida et al. Comput Biol Med; 2024;182:10907).
Computational fluid dynamics (CFD) simulations offer a powerful way to analyze blood flow in cases of suspected CoA. The simulations enabled the team to observe how changes in the aortic arch shape influence blood circulation, focusing on factors like wall shear stress and flow direction. By modeling these interactions, CFD highlights patterns, such as retrograde flow or stress imbalances that are commonly linked to CoA. These findings can be tied to specific anatomical features, helping to identify risk factors that might not be apparent from imaging alone. Additionally, the ability to isolate and manipulate variables like ventricular output in a controlled virtual environment provides valuable insights into how fetal cardiovascular systems adapt to structural changes. This approach is particularly useful for studying subtle hemodynamic variations that might differentiate true CoA cases from false positives.
“Simulation helped explain why a vessel might have a defect like coarctation,” says Dr. de Vecchi. “This is an interplay between flow and anatomy. It's how the flow itself can change anatomy during development because of the shear on the walls and the volume of flow that can dilate the vessel, make it grow more, and so on. But at the same time, obviously the shape of a vessel will change the flow. And it's quite difficult to understand if it's driven by the flow, if it's driven by anatomy, and at what stage.”
Incorporating these technologies with advanced imaging reconstruction techniques ensures accurate representation of the fetal heart, even in challenging imaging conditions. The ability to digitally manipulate and analyze anatomical data in 3D not only improves diagnostic precision but aids in understanding the structural and flow changes associated with CoA, paving the way for more reliable risk assessment before birth.
Creating a Better Risk Assessment
The study that the team conducted was so successful that it actually influenced a new set of risk assessment parameters at Evelina Children’s Hospital. Typically, doctors review the ultrasound and make an educated prediction based on standard cardiac measurements. However, these measurements are known to be poor predictors of neonatal CoA, meaning that many newborn babies end up separated from their families, receiving unnecessary treatment and monitoring in intensive care until the condition can be reliably excluded after birth.
Using the roughly 200 patient-specific computer models that the team created, they trained a classifier to extract parameters from the digital twins and produce a percentage of likelihood of CoA before the baby is born.
“It basically automatically extracts a number of parameters from fetal cardiac MRI scans, and then, using linear discriminative analysis, presents us with risk of early postnatal coarctation,” says Dr. Lloyd. “We can visualize this result on an axis of disease that goes from healthy to CoA, giving us an objective risk score to help decide whether the baby will need early neonatal surgery.”
Clinicians at Evelina Children’s Hospital can now run these reports simply and efficiently with a single click, and use the percentages to provide additional data to help in diagnostic decision-making. The team has already integrated these pipelines as part of the assessment of patients having MRI scans for suspected CoA scanned at St. Thomas’, and their performance is being evaluated alongside existing clinical tests.
Future Prospects and Research Directions
King's College London is working to refine digital twin models for improved detection and management of CoA. Future imaging studies during pregnancy could even provide insights into how the condition develops in fetal life, by simulating the effects of mechanical forces, such as pressure and flow patterns, on vessel growth to better understand the condition's underlying mechanisms.
Overall, the team believes these advancements could enable earlier, more precise interventions, improving neonatal outcomes and reducing uncertainty, stress, and anxiety on expectant mothers and their families. Continued collaboration across clinical, computational, and research fields is expected to further optimize these technologies, paving the way for broader adoption of advanced simulation tools in neonatal cardiovascular care.
Learn more about how Ansys, part of Synopsys, can help with your cardiovascular applications.
Just for you. We have some additional resources you may enjoy.
“We started to see some features in the flow that only happen in true CoA but not in false positives. Fluent really helped us understand the mechanism at play.”
— Dr. Adelaide de Vecchi, head of the Research Department of Digital Twins for Healthcare at King's College London
The Advantage Blog
The Ansys Advantage blog, featuring contributions from Ansys and other technology experts, keeps you updated on how Ansys simulation is powering innovation that drives human advancement.