ANSYS BLOG
April 24, 2020
How do Adhesives Work? The Science of Stickiness
Pressure sensitive adhesive tapes can be useful in a variety of situations from creating artwork, to sealing packages to saving the lives of Apollo 13 astronauts. Though these products are ubiquitous, many of us still wonder: How do adhesives work and what makes things sticky?
The science behind the conditions under which things stick is fascinating. It’s why so many of us played with tape, gum and glue when we were young.
For some, that curiosity expanded, evidenced by the amount of active research into stickiness to advance the chemical, biomedical and electronics industries.
For instance, read our recent publication in Scientific Report at Nature Research: Two-Dimensional Finite Element Analysis of Elastic Adhesive Contact of a Rough Surface.
So, based on that research, let’s answer this question once and for all: Why are things sticky?
How do adhesives work and why are things sticky? Many of us have wondered about this.
How do Adhesives Work?
The stickiness of objects depends on the ability of its surface to make intimate contact with other surfaces. If the surfaces make enough contact, their interatomic forces act like Velcro and bind the surfaces together.
Why does one end of the tape stick to the box and the other doesn’t stick to the hand?
It’s because of the addition of a compliant polymer to one side of the tape.
For stiff materials, like metals, having a surface roughness of a nanometer removes the effects of stickiness. More compliant materials, like rubbers and polymers, can remain sticky as long as the surface roughness measures less than a micrometer.
For adhesive tapes, a very thin layer of highly compliant polymer resides on one side. It enables the tape to make intimate contact with various dry surfaces. That explains why the tape sticks to our hands despite the rough ridges in our palms.
The opposite side of the tape, the one without the compliant polymer, exhibits stiffness that cannot overcome the roughness of our hands. As a result, it fails to form intimate contact and remains non-sticky.
Engineers Need to Understand What Makes Things Sticky
To better understand the science behind stickiness, and its potential benefits, engineers often look to objects and creatures in nature.
For example, gecko lizards have evolved padding under their feet that stick to various dry surfaces. This enables them to climb walls and glass panes with ease.
Some plants, like the lotus, have evolved surface textures that minimize the effect of stickiness (or wettability). This enables them to repel water droplets.
Better understanding of these phenomena could help improve the quality of daily life. For instance, mimicking the lotus plant could help engineers develop medically sterile surfaces that prevent moisture and bacterial growth.
Additionally, research into reducing, or increasing stickiness could help engineers develop better sensors, food science and manufacturing technologies.
By studying the stickiness of nature, engineers can learn how to make better products.
How to Simulate Stickiness
Engineers that study how to control stickiness require complex computational models that mimic the interatomic forces that cause surfaces to stick to each other. These models must also be able to accurately represent the texture, or profile, of the surfaces down to the nanoscale.
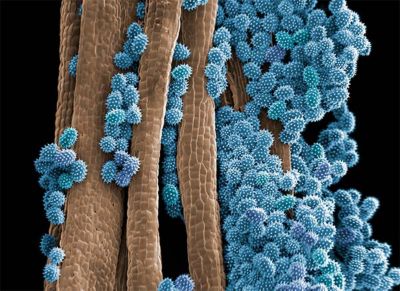
A scanning electron microscope (SEM) image of pollen sticking to a surface. Pollen has textured spikes that enable it to adhere to the hairs of insects.
These simulations can be produced using the highly nonlinear, and large model capabilities of Ansys Mechanical. Engineers can then utilize user programmable features (UPFs) to customize the system’s materials, loads and interfacing bodies to study how objects connect. Mechanical’s ability to model complex interaction between surfaces on the nanoscale, with a large number of elastic instabilities, makes the problem manageable.
Using these simulations, engineers enhance or minimize the effect of surface adhesion by controlling the:
- Surface profiles
- Compliance of the interfacing bodies
- Material selections
This helps them study phenomena like stiction and friction — a common challenge in the development of sensors and bioimplants.
If you are still wondering: Why are things sticky? Learn how to produce models to test the phenomena yourself, read our recent publication in Scientific Report at Nature Research: Two-Dimensional Finite Element Analysis of Elastic Adhesive Contact of a Rough Surface.