ANSYS ADVANTAGE MAGAZINE
OCTOBER 2021
Each year, thousands of people die as a result of aortic aneurysms — balloon-like bulges caused by weaknesses in aortas, the large artery that sends blood from the heart through the vascular system. If an aneurysm remains undetected, the aorta can eventually tear or rupture, causing uncontrolled internal bleeding.
When an aneurysm is detected before it ruptures, thoracic surgeons can intervene by repairing the aorta and eliminating the aneurysm. However, repair surgery comes with risks. If surgeons use conventional open-surgery approaches, up to 12% of patients may not survive the procedure.

Aneurysm model
A newer and less invasive technique, endovascular aneurysm repair (EVAR), has a much lower mortality rate. In this procedure, surgeons make a small incision in the groin, through which they insert a stent graft and thread it upward until it reaches the aneurysm. There, it is positioned optimally to reinforce the section of weakened tissue.
The greatest challenge of an EVAR procedure is that surgeons are operating blindly, without a detailed understanding of each patient’s unique vascular geometry. While imaging tools help guide the insertion and placement of the stent, the process is characterized by real-time discovery and adaptation. Surgeons have little advanced knowledge of the patient’s vascular system in three dimensions because imaging tools like computed tomography (CT) scans only reveal two dimensions.
Simulation Provides a Clear, Patient-Specific View
Professor Jean-Philippe Verhoye is a leading global advocate for using biomedical simulations of individual patients’ vascular systems to inform surgery and produce better outcomes.
Prof. Verhoye is head of cardiac and vascular thoracic surgery at Rennes University Hospital, France. He is also the president of the French Society of Thoracic, Cardiac and Vascular Surgery, and a member of the European Society of Thoracic, Cardiac and Vascular Surgery, as well as former secretary of the French College of Cardiac Surgery. He has published 190 articles and two books on cardiac and vascular surgery topics.
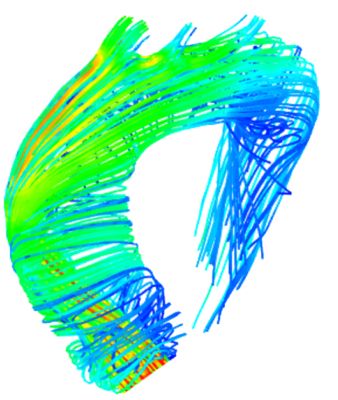
Aortic flow simulation
“By using routinely available images like CT scans and other clinical data to generate 3D virtual models of the vascular system, surgeons can ‘practice’ their EVAR procedures in advance, using the actual patient’s specific geometry,” says Prof. Verhoye. “Since no two patients’ bodies or vascular systems are alike, surgeons can optimize the process of threading and positioning the stent on an individual basis.
“Simulation allows the creation of a kind of digital twin — or avatar — of a given human body,” explains Prof. Verhoye. “We can work on that personal digital avatar and perfect our surgical techniques without any degree of risk exposure for the patient.”
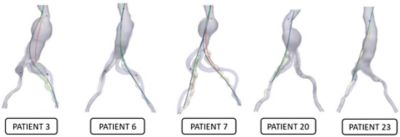
These simulations of endovascular aneurysm repair (EVAR) surgery, modeled using Ansys software, show the very different vascular geometries of five patients with an aortic aneurysm. Surgeons at Rennes University Hospital, France, were able to simulate the insertion of an aortic stent in advance (path shown in black) for an optimal, error-free outcome (path shown in red).
From Emerging Concept to Everyday Practice
For more than seven years, Prof. Verhoye’s cardiac and thoracic surgery team at Rennes University Hospital has been perfecting the use of patient modeling via Ansys LS-DYNA prior to EVAR procedures. “We have a dedicated team of researchers who perform the meshing and modeling, and they work closely with the surgical team,” he notes. The resulting model not only reflects the overall vascular geometry, but also the distance between the aorta and the spinal cord ― a critical boundary condition — and the properties of artery walls.
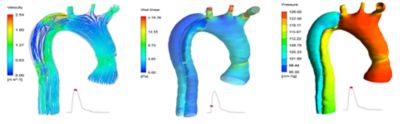
In modeling a specific patient’s vascular system, Ansys software accurately replicates the overall organ geometry, as well as key properties such as calcification or thrombosis of the artery walls.
“Ansys has collaborated with us to ensure that the available data, including CT and MRI (magnetic resonance imaging) scans, feeds into the software to automatically produce a geometrically correct, finely meshed model,” Prof. Verhoye continues. “Ansys has also helped us develop some specialized expertise, such as understanding what kinds of tissue deformation occur when the stent is inserted.”
Prof. Verhoye and his team use the Ansys-enabled modeling approach during more than 100 successful EVAR surgeries each year. “It has taken us years to get from the initial idea to practical application,” he states, “but today pre-surgery modeling is a routine procedure for aortic aneurysm patients at Rennes.”
Patient-specific modeling provides Prof. Verhoye’s team with three distinct capabilities. “First, simulation allows us to accurately visualize the interior of a patient’s body, without opening the thoracic cavity,” he says. “That in itself is revolutionary. But it also enables us to confidently predict outcomes and then communicate about surgery challenges on a case-by-case basis. We go into surgery with a shared expectation of what will happen. That’s invaluable.”
Modeling a More Innovative Future
Not only is Prof. Verhoye performing simulation-driven surgery himself, but he is also training the next generation of surgeons in these advanced techniques. “I think in 10 to 15 years, pre-surgery modeling will become an industry-standard procedure,” he predicts. “But we will have to undergo massive clinical trials before regulatory and insurance company approvals are gained on a large scale. Until then, we are building our own capabilities here and establishing best practices.”
Beyond approvals, Prof. Verhoye points out that there are cultural and financial obstacles standing in the way of faster adoption. “We are lucky that Rennes understood the concept of simulation and its potential benefits very quickly,” Prof. Verhoye points out. “We have been able to invest in proving the idea. Not every university or hospital is open to supporting this kind of radical innovation.
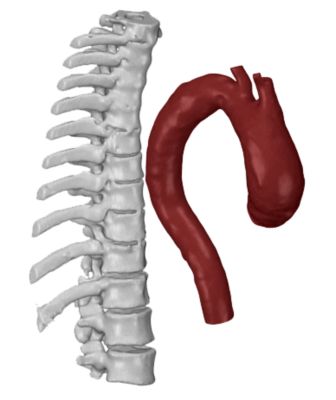
A model represents the distance between the aorta and the spine.
“We have also been given the freedom to assemble a simulation team that really understands our challenges as surgeons,” Prof. Verhoye continues. “Often there is a disconnect between clinicians and engineers, but we have been able to overcome that challenge by living, eating and sleeping together. We speak the same language now. We are equally devoted to applying the best-available technologies to improve patients’ outcomes and save lives. That is how real biomedical innovation takes place.”
The model also shows properties of artery walls.
现在就开始行动吧!
如果您面临工程方面的挑战,我们的团队将随时为您提供帮助。我们拥有丰富的经验并秉持创新承诺,期待与您联系。让我们携手合作,将您的工程挑战转化为价值增长和成功的机遇。欢迎立即联系我们进行交流。