Optimize Cardiovascular Treatments with Simulation
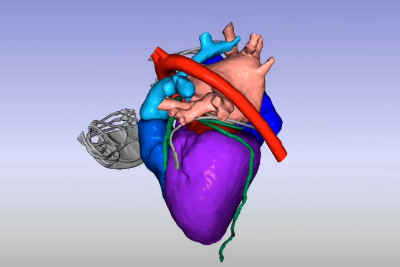
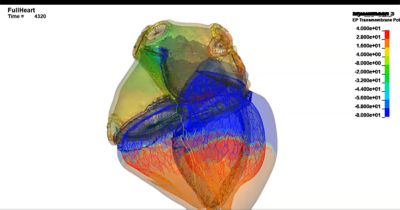
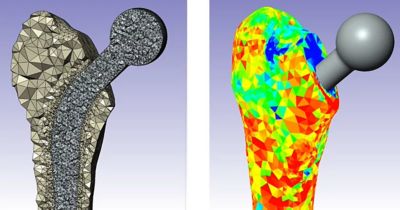
Understanding the fluid-structure-electrophysiology interactions (FSEI) of the heart is essential for treating conditions such as heart failure, arrhythmias, and valve diseases, and for determining how medical devices and pharmaceuticals affect the heart’s behavior. The healthcare industry uses in silico technologies, such as computational modeling and simulation (CM&S), artificial intelligence (AI), and digital twins, to better understand these behaviors and develop life-saving devices and treatments for cardiac pathologies. By creating validated, multiphysics models of the heart, cardiac devices, and drug interactions, innovators can design, optimize, and test safe cardiovascular treatments.